Notes-(01)
Completion requirements
(d) describe sp3 hybridisation, as in ethane molecule, sp2 hybridisation, as in ethene and benzene molecules, and sp hybridisation, as in ethyne molecule
(e) explain the shapes of, and bond angles in, the ethane, ethene, benzene, and ethyne molecules in relation to σ and π carbon-carbon bonds
4. sp2 Hybridization
4.2. Shape and Bond Angle
o Electrons repel one another
o \(sp^2\) hybrid orbitals are placed as far apart as possible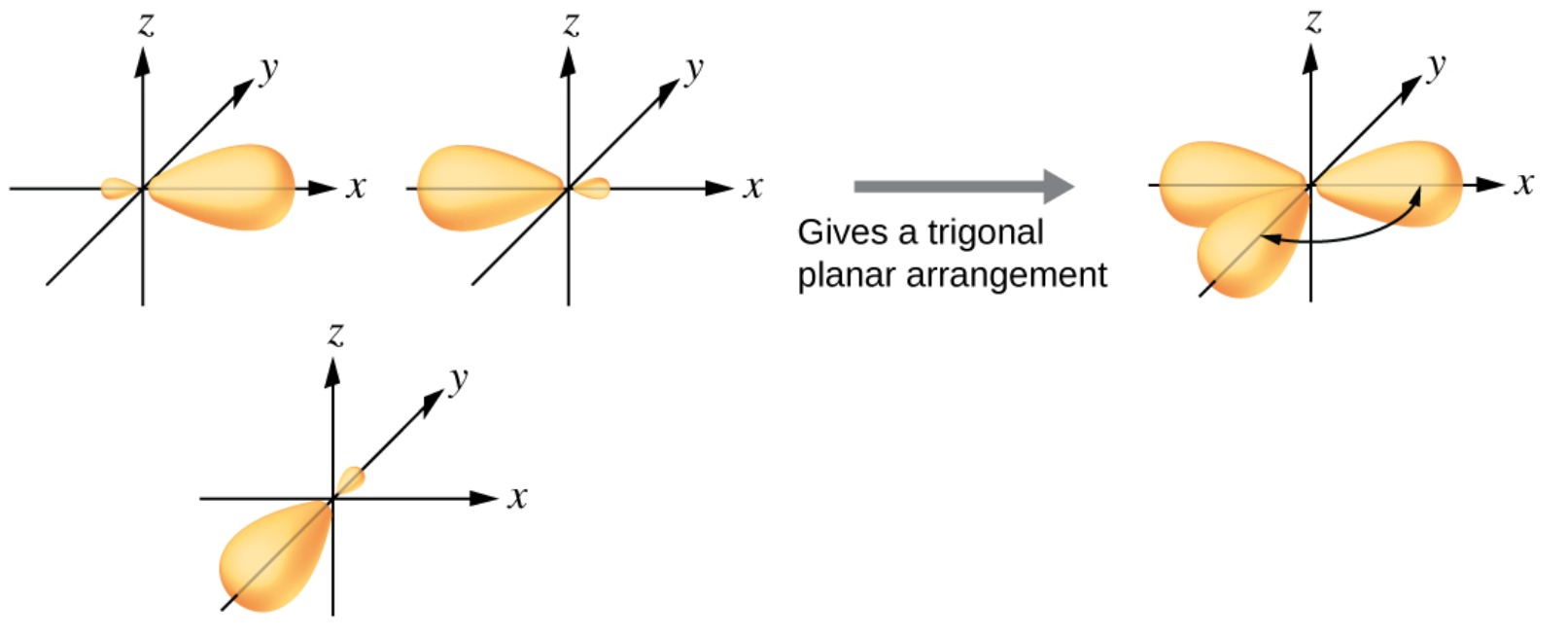
Shape: Trigonar planar
Bond Angle (Around C): 120°